
XEOMIN® is injected directly into the parotid and submandibular salivary glands
The following videos give guidance for injection with the use of ultrasound or surface anatomical landmarks.
XEOMIN® (incobotulinumtoxinA) is indicated for the treatment of chronic sialorrhea associated with neurological disorders in adults. Please refer to the Product Monograph for complete dosing and administration information.


Injection Training
Anatomic Landmark Guidance
Ultrasound Guidance
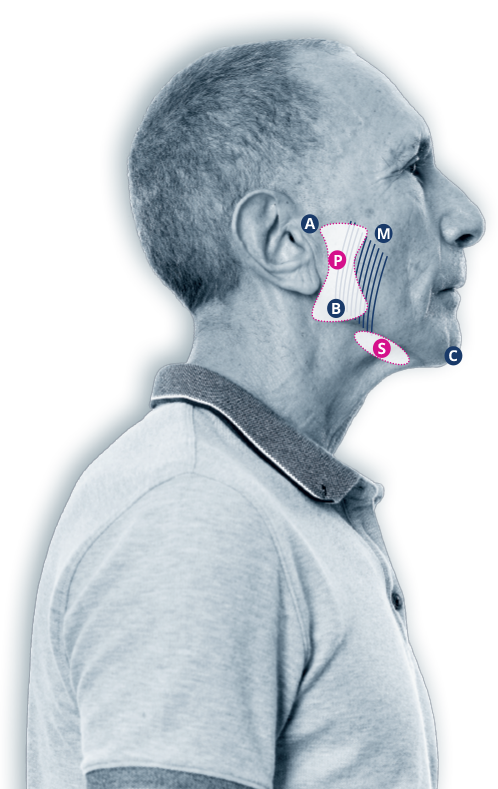
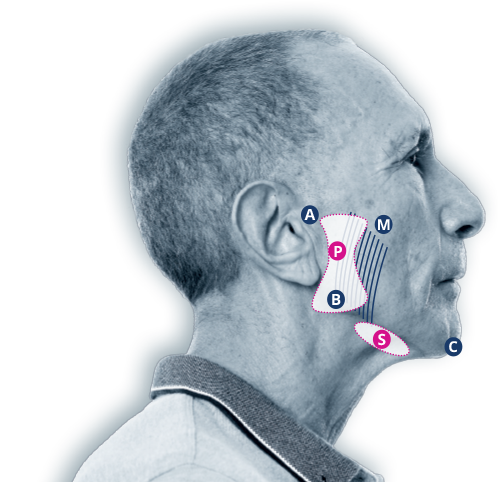
- Parotid gland(s)
30 U Injection - SUBMANDIBULAR GLAND(S)
20 U Injection
- TRAGUS
- MANDIBLE ANGLE
- TIP OF CHIN
- MASSETER
Recommended dosage by injection location
XEOMIN® is injected into the parotid and submandibular glands on both sides (i.e., 4 injection sites per treatment session). The injection site should be close to the centre of the gland.
A reconstituted solution with a concentration of 5 units / 0.1 mL should be used. Reconstitute the 100 units with 2.0 mL of a sterile 0.9% sodium chloride solution.
The timing for repeat treatment should be determined based on the actual clinical need of the individual patient, and no sooner than every 16 weeks.
| GLAND(S) | UNITS PER SIDE | TOTAL |
|---|---|---|
| Parotid gland(s) | 30 Units | 60 Units |
| Submandibular gland(s) | 20 Units | 40 Units |
| Total | 50 Units | 100 Units |

XEOMIN® HAS 3 OTHER INDICATIONS
XEOMIN® is also indicated:
- for the treatment of hypertonicity disorders of the 7th nerve such as blepharospasm including benign essential blepharospasm and hemifacial spasm in adults.
- to reduce the subjective symptoms and objective signs of cervical dystonia (spasmodic torticollis) in adults.
- for the treatment of upper limb spasticity associated with stroke in adults.
For more information:
Please consult the Product Monograph at https://www.merzcanada.com/en/xeomintherapeutic.html for important information relating to contraindications, warnings, precautions, adverse reactions, interactions, dosing, and conditions of clinical use, which have not been discussed in this piece. The Product Monograph is also available by calling 1-877-336-4008.
Reference: Merz Pharma Canada. XEOMIN® Product Monograph.


